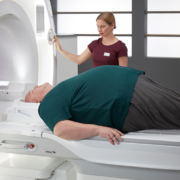
High-end MRI scanner adapts automatically to individual anatomical and physiological characteristics
Magnetom Vida, the new high-end 3-Tesla MRI scanner with BioMatrix
technology from Siemens Healthineers, was launched to the public at University Hospital Tübingen, where the first system is installed. It has been undergoing clinical tests in the hospital’s Department for Diagnostic and Interventional Radiology since December 2016.
Magnetom Vida is the first scanner equipped with BioMatrix, a brand-new, innovative scanner technology that addresses inherent anatomical and physiological differences among individual patients, as well as variability among users. Magnetom Vida and BioMatrix allow users to meet the growing demand for MR imaging, perform the full range of routine as well as complex examinations, and deliver robust results for every patient. Furthermore, the scanner also makes MRI more cost-effective by reducing rescans and increasing productivity. High-precision imaging means that radiologists can deliver essential and robust information to choose the right treatment for each patient every time. Siemens Healthineers, in collaboration with its customers, is playing an important role in taking healthcare forward in the development of precision medicine.
Siemens Healthineers has been developing this disruptive and innovative BioMatrix technology for over five years. Its introduction represents a further advance in MRI imaging as well as the next level of automation and patient centricity.
High image quality and efficient workflows – regardless of user or patient
Due to high levels of exam variability, MRI is often considered to be one of the most complex medical imaging modalities. Physiological and anatomical differences between patients as well as different experiences levels in users contribute to this unwanted variability. This frequently is a source of errors, rescans, and inefficient workflows in MR imaging, making it all the more important that MRI scanners deliver reliable and reproducible image data irrespective of the patient being examined or the person operating the system. This issue is precisely addressed with the new BioMatrix technology.
BioMatrix sensors in the table automatically track a patient’s respiratory pattern, giving users insights into a patient’s individual ability to hold his or her breath during the scan. This allows the user to select the optimal exam strategy, while also saving time during the examination. BioMatrix tuners can help avoid rescans, which represent a major burden on productivity as well as a driver of additional costs in radiology. In cervical spine examinations, for example, this feature uses intelligent coil technology to automatically set the optimal scan parameters based on the individual patient anatomy, all without any additional user interaction. BioMatrix tuners also improve the quality and reproducibility of whole-body diffusion. Precise control of scan parameters in real-time to match the individual patient anatomy makes it possible to avoid distortions, which can render diffusion imaging non-diagnostic, especially in 3 Tesla MRI. Innovative interfaces also help ensure a consistently high examination quality, accelerating workflows, and improving quality of care. BioMatrix Interfaces accelerate the scanning process by up to 30 percent. Automated patient positioning based on intelligent body models automatically moves the patient table to the correct scan position. An intuitive touchscreen user interface integrated onto the scanner allows for one-touch positioning. A new, easy-to-move motorized patient table further simplifies examinations, especially for adipose, immobile, and trauma patients.
Magnetom Vida is the first system to be equipped with the new BioMatrix technology, designed to tackle the challenges of variability and thereby, reduce unwanted variability in MRI examinations. It will help users achieve fewer rescans, predictable scheduling, and consistent, high-quality personalized examination results.
The ability to provide consistent and reproducible quality regardless of the individual patient and user will help reduce rescans, which can be a great financial burden for healthcare institutions. As publications have shown, rescans can account for up to €100,000 per year and system in additional costs.
Professor Konstantin Nikolaou, Medical Director of the Department of Diagnostic and Interventional Radiology at University Hospital Tübingen considers Magnetom Vida to be part of the general trend toward precision medicine: “To provide our patients with individual therapies, we need every piece of information available. When it comes to imaging, this means that we need robust, standardized, and reproducible image data that are always of the same quality regardless of the patient or user. Only then we can compare results and link them with additional information, such as data from laboratory medicine or genetics,” says Nikolaou, referring to the clinical validation of the new MRI scanner in his department. “Magnetom Vida gives us this data quality and comprehensive image information so that we can choose the right kind of personalized therapy and evaluate it – to see, for instance, how a patient responds to chemotherapy before tumour removal. This MRI scanner along with BioMatrix technology is the perfect fit for our current medical approaches, and is helping us on our way to quantitative radiology,” says Nikolaou.
Faster scans with very high patient comfort
Magnetom Vida has another major advantage: “We can examine sick patients faster with Magnetom Vida,” says Professor Mike Notohamiprodjo who, as head of MRI at University Hospital Tübingen, works intensively with the new scanner. “The scanner offers the highest degree of patient comfort with the performance of a research system, which speeds up our workflows,” he says. As examinations in Tübingen show, the new scanner decreases measurement times for musculoskeletal and prostate imaging compared to previous MRI systems. What is more, it does so with significantly improved image quality: “The signal-to-noise ratio in the clinical images is up to 30 percent higher than with systems from the previous generation,” says Notohamiprodjo.
While this is partly due to BioMatrix technology, it is also a result of the diverse insights that developers at Siemens Healthineers gathered from intense fundamental research and close customer collaborations. Key learnings from the development of a 7-Tesla research MRI system translated into a new 3-Tesla magnet design. Magnetom Vida’s all-new system architecture offers extremely high performance and unmet long-term stability – without requiring any more space than previous clinical systems. The new scanner’s 60/200 XT gradient system provides over 2.7 megawatts of power, making it the most powerful commercially available gradients in a 70-centimeter bore scanner. And, thanks to a very large field of view (55x55x50 cm), Magnetom Vida can also cover larger body regions in one step, such as full coverage abdominal exams.
The result is a great increase in productivity for routine examinations of the brain, spine, and joints – from correct patient positioning at the touch of a button to transferring the clinical images to the PACS archiving system. This is made possible by the GO technologies, which automate and simplify workflows from the start of the scan right through to the quality control of the image data. A new user interface allows not only for automated acquisition and processing, but also for more advanced post-processing applications to run at the scanner. With spine examinations, for instance, GO technologies reduce the time needed by about a fifth. This means that a department could carry out four additional spine examinations per day and per system. Given the decline in reimbursement rates, this is of great value to many radiological institutes.
Broader patient groups and new clinical growth areas
The system also allows customers to access additional clinical growth fields – for instance, by serving patient groups that were previously deemed unsuitable for MRI due to issues such as cardiac arrhythmias, excess weight, or health problems that prevent them from actively supporting the scan. With the introduction of Magnetom Vida, Siemens Healthineers expands its Compressed Sensing applications – which can make MRI scans up to ten times faster – to cover more body regions. It features Compressed Sensing Cardiac Cine, which allows free-breathing cardiology examinations (even when using contrast medium for comprehensive tissue characterization). Now, Compressed Sensing Grasp-Vibe, which enables dynamic, free-breathing liver examinations in one comprehensive scan by the push of button and for every patient, is also available. Until today, in contrast, dynamic liver imaging required four steps with exhausting breath-holds and complex timing. Grasp-Vibe technology also makes the post-processing of liver images significantly faster. During the studies he carried out in Tübingen, Professor Notohamiprodjo found that post-processing times fell from 20 to just four minutes.
Magnetom Vida even simplifies whole-body scans, which are currently particularly challenging, because they have to cover multiple scan sections and demand highly trained users. A new special technology, the Whole-Body Dot Engine, allows these difficult scans to be carried out in predictable time slots, as short as 25 minutes, with very high quality. This is accomplished through intelligent automation. The planning and execution of the scan requires only a few simple clicks. Providing high-quality diffusion weighted imaging is important for whole body exams; Magnetom Vida, with its BioMatrix Tuner technology, can deliver this distortion-free. Combined also with its strong 60/200 gradients and a large homogeneous field of view, Magnetom Vida makes whole-body examinations simple to perform, reproducibly, and with very high-quality. This is a major advantage, particularly when treating oncology patients, such as those with multiple myeloma, where guidelines have recently been moving toward whole-body MRI scans for therapy control.
Magnetom Vida offers not only numerous clinical advances, but also a number of improvements in energy consumption. These help to lower the total cost of ownership of the system over its entire life-cycle. Technologies such as Eco-Power provide an intelligent control of power-hungry components by switching them off when they are not needed for longer periods of time. The result is a MR scanner that consumes 30 percent less energy than the industry average for 3-Tesla scanners, as reported by the European Coordination Committee of the radiological, electromedical and healthcare IT industry (COCIR).