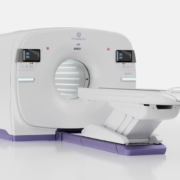
GE HealthCare receives CE mark for 128cm total body PET/CT
GE HealthCare has secured CE mark approval for its Omni 128cm total body positron emission tomography/computed tomography system, designed to advance cancer diagnosis, staging and therapeutic planning through enhanced molecular imaging capabilities. The next-generation platform addresses growing demand for whole-body PET/CT imaging as global cancer rates are projected to increase 77 per cent by 2050.