Researchers develop safe bioink for 3D printing artificial organs
The Korea Institute of Science and Technology (KIST) has developed a new type of bioink material for 3D bioprinting that can be used to create artificial tissues and organs. The new bioink is based on a temperature-sensitive poly(organophosphazene) hydrogel.
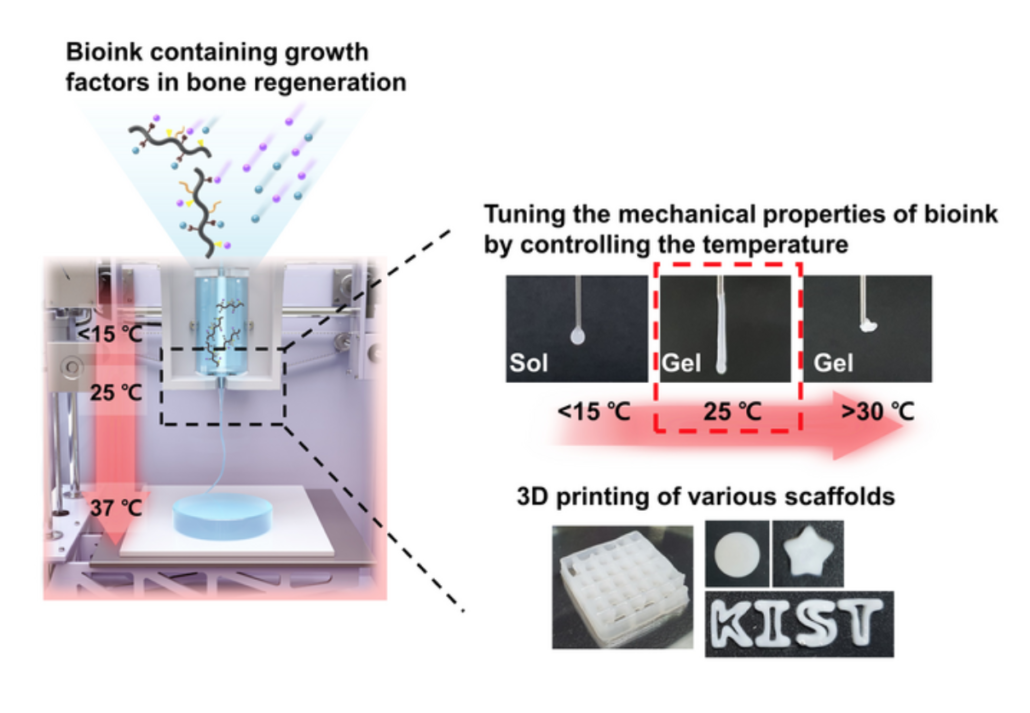
Tuning mechanical properties of bioink according to temperature and 3d scaffold printing
The hydrogel exists in a liquid form at low temperatures and changes to a hard gel at body temperature. This enables the regeneration of tissues by temperature control only, without chemical crosslinking agents or UV irradiation and the manufacture of a three-dimensional scaffold with a physically stable structure, which minimizes the possibility of immune adverse effects in the human body.
The bioink can also interact with growth factors to enhance tissue regeneration, and it can be tailored to different types of tissues and organs.
The research team fabricated the 3D scaffold by printing it with a 3D bioprinter using bioink containing transforming growth factor beta 1 (TGF-β1) and bone morphogenetic protein-2 (BMP-2), which are required for cell infiltration and bone regeneration. They conducted an experiment by implanting it into a damaged bone in a rat. This resulted in cells from the surrounding tissue migrating into the scaffold and regenerating the damaged bone to a normal tissue level.The implanted 3D scaffold slowly biodegraded in the body over 42 days.
The development of this new bioink is significant because commonly used hydrogel-based bioinks can cause cytotoxicity due to the chemical crosslinking agent and ultraviolet light used to connect the molecular structure of photocuring 3D-printed bioink. The new bioink developed by KIST eliminates this risk and allows for the creation of a three-dimensional scaffold with a physically stable structure that minimizes the possibility of immune adverse effects in the human body.
The technology has already been transferred to NexGel Biotech Co., Ltd. for commercial development, and the team is conducting follow-up research to apply it to the regeneration of other types of tissues and organs.
Overall, this new bioink technology has great potential for use in the development of artificial organs and tissues for patients suffering from accidental injuries and chronic diseases.