Philips SmartCT 3D image acquisition for Azurion system gets FDA clearance
Royal Philips, has received U.S. FDA 510 (k) clearance for its Philips SmartCT application software. SmartCT is a key component of Philips Image Guided Therapy System – Azurion – providing interventionalists with CT-like 3D images (Cone Beam CT) to support diagnosis, therapy planning, treatment and follow-up for interventional radiology procedures.
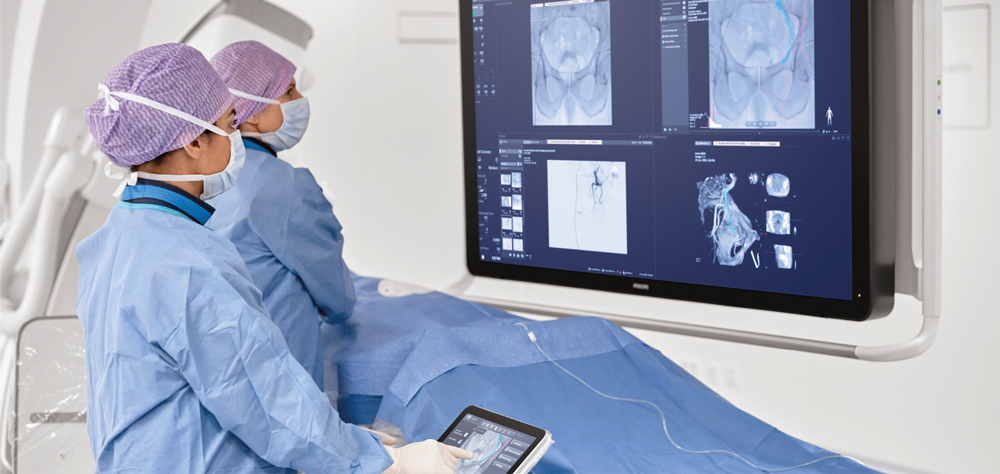
Bringing intuitive touchscreen control of advanced 3D image acquisition, visualization, vessel/organ segmentation, and quantitative measurements to the table-side, within the interventional lab’s sterile zone, SmartCT helps enhance clinical confidence, smooths workflows, and increases productivity. It includes software applications for angiography, neurology, soft-tissue imaging, and guidewire/catheter navigation, supporting a wide range of procedures such as the treatment of aneurysms, vascular diseases and liver tumours.
Philips’ latest Azurion image-guided therapy platform integrates essential lab systems and tools needed for complex interventional procedures into an uncluttered laboratory environment in which interventionalists can focus on treating the patient rather than being distracted by the technology. SmartCT brings total control of the Azurion platform to a touchscreen tablet situated alongside the interventional radiology table. This eliminates the need for clinicians to leave the sterile field and step into an adjacent control room, as well as supporting faster and better-informed decision making.
A key part of our image-guided therapy strategy is to combine high-quality, low X-ray dose imaging with a superior user experience that allows interventional radiologists to diagnose and treat patients as part of smoother, safer and less interrupted workflows.
Ronald Tabaksblat, General Manager Image Guided Therapy Systems at Philips, commented: “Philips SmartCT is a major step forward in 3D imaging, enhancing confidence in the interventional suite and supporting key elements of the quadruple aim of better patient outcomes, enhanced patient and staff experiences and lower cost of care.”
Prof. Marc Sopval, Interventional Radiologist at Hôpital Européen Georges Pompidou AP-HP in Paris, France, commented: “With the new SmartCT interface we can go into more detail more quickly and safely, with fewer staff in the room. SmartCT has brought Cone Beam CT to life in everyday practice: all the tools and guidance capabilities are used by the entire team each day.”
With SmartCT, users are guided through the image acquisition process and can review and interact with the acquired CT-like 3D images on the system’s table-side touch screen module using 3D visualization and measurement tools. These tools have been designed to support procedures in a range of clinical domains, including neurology, oncology, and cardiovascular procedures, and feature intuitive two-point distance measurements on 3D images, the ability to remove structures from the images that obstruct the region of interest, and the ability to select and store optimum projection angles for recall during procedures.
Philips SmartCT image acquisition, visualization and measurement software is an integral part of the next-generation Philips Image Guided Therapy System – Azurion – which was launched in September 2020, marking an important step forward in optimizing clinical and operational lab performance and expanding the role of image-guided interventions in the treatment of patients. Azurion has achieved rapid global adoption, reflecting the accelerating trend toward minimallyinvasive surgery thanks to its benefits in terms of reduced patient trauma, shorter recovery times and hospital stays, and lower healthcare costs.
For more information, visit: https://www.usa.philips.com/healthcare/product/HCNCVC846/smartct-3dvisualization-and-measurement-solution