Philips launches new compact ultrasound system
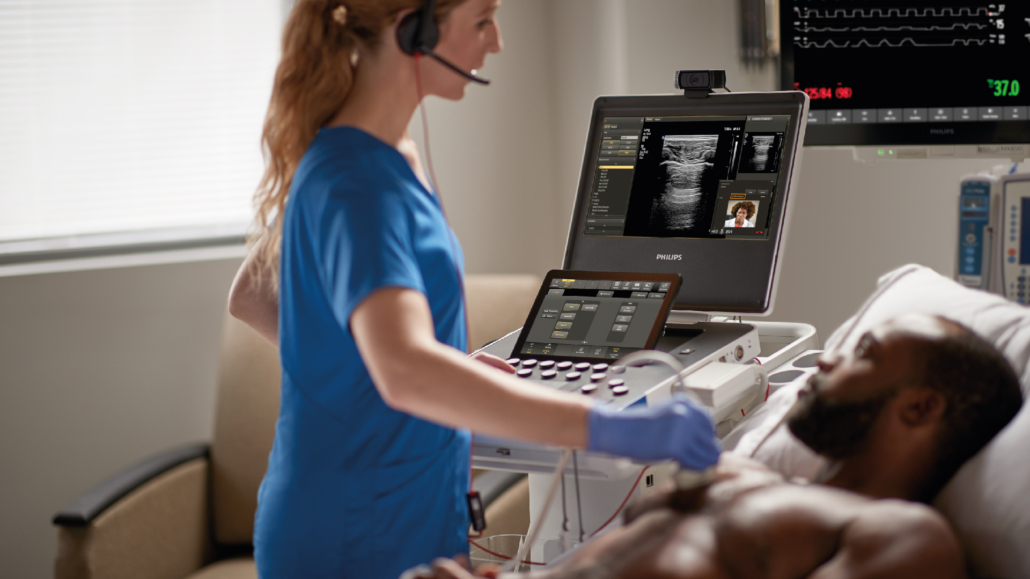
Philips has launched globally their next-generation compact portable ultrasound solution, the Compact 5000 Series. This new ultrasound is designed for portability and versatility without compromising image quality or performance and aims to bring the diagnostic quality associated with premium cart-based ultrasound systems to more patients.
Replicating the intuitive user interface and workflow of Philips’ cart-based systems EPIQ Elite and Affiniti, and offering full compatibility with these systems’ transducers, the Compact 5000 Series supports an easy learning curve for users. Designed for shared service capabilities across specialties including cardiovascular, obstetrics and gynaecology, point of care, and general imaging, the Compact 5000 Series system helps meet the needs of multiple clinical segments. The Compact system can be configured with an optional battery allowing for 2.5 hours of scanning time and faster power-up capability to quickly ready the system at the patient’s side. Additionally, the new system features fewer hard keys on the console and a fully sealed control panel for easy disinfection and cleaning protocols.
“Improving patient outcomes and the patient experience means providing clinicians with our best diagnostic tools available and making advanced technology available wherever the patient is located,” said Jeff Cohen, general manager of Ultrasound at Philips. “With the patient at the centre of everything we do, we built the Ultrasound Compact System 5000 Series to perform as a highly versatile portable ultrasound system to be used in multiple care settings for many different types of exams without compromising image quality, diagnostic ability, or clinical workflow.”
This newest system also features ‘Philips Collaboration Live’ secure real-time telemedicine software, with the ability to call on additional clinical expertise via secure voice, text, screen sharing, and video streaming from anywhere with a mobile or internet connection. Collaboration Live even allows a remote user to control the unit’s settings to acquire diagnostic quality images, reducing the need for follow-up scans and patients having to travel to a central hospital facility.
The Compact 5000 Series recently received U.S. FDA 510(k) clearance and is pending CE mark.
For more information, visit: https://www.philips.com/global
Digital issue: Please click here for more information