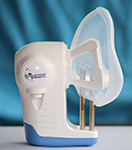
Pediatric disease breathalyser
Owlstone Medical, a diagnostics company, has developed and received CE mark approval for a pediatric version of the company’s disease breathalyser, ReCIVA. The marking extends the scope of breath testing in early stage diagnostics and therapy response to include children and in particular, the difficult to manage group of child asthma patients. Both the adult and pediatric versions of the breathalyzer are now being used in EMBER (East Midlands Breathomics Pathology Node), a