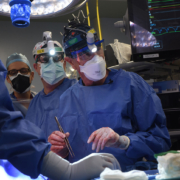
Patient receives pig heart in world-first transplant
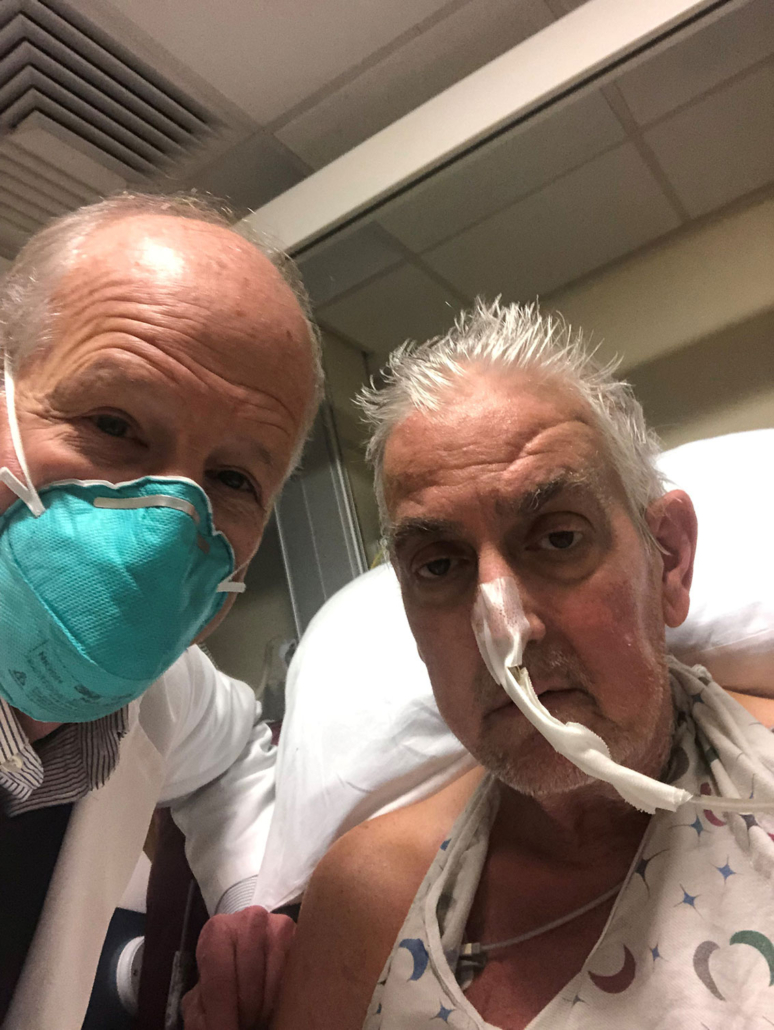
Bartley P. Griffith, MD and the patient, David Bennett — University of Maryland School of Medicine
In a major medical breakthrough, a man in the United States has received a heart from a genetically modified pig in a successful transplant.
The 57-year-old man, David Bennett, who was not eligible for a routine heart transplant, opted for the experimental surgical procedure, saying: “It was either die or do this transplant. I want to live. I know it’s a shot in the dark, but it’s my last choice.”
The eight-hour surgery took place on Friday 7 January and at the time of this publication, the patient was reportedly doing well.
The historic surgery was conducted by a team lead by Bartley P. Griffith, MD, and Muhammad M. Mohiuddin, MD, at the University of Maryland Medicine.
This experimental transplant procedure shows that a genetically-modified animal heart can function like a human heart without immediate rejection by the body and has major implications for the future of transplant surgery.
The U.S. Food and Drug Administration granted emergency authorization for the surgery on New Year’s Eve through its expanded access (compassionate use) provision. It is used when an experimental medical product, in this case the genetically-modified pig’s heart, is the only option available for a patient faced with a serious or life-threatening medical condition.
Dr Griffith, who is the Thomas E. and Alice Marie Hales Distinguished Professor in Transplant Surgery at University of Maryland School of Medicine (UMSOM), commented: “This was a breakthrough surgery and brings us one step closer to solving the organ shortage crisis. There are simply not enough donor human hearts available to meet the long list of potential recipients. We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future.”
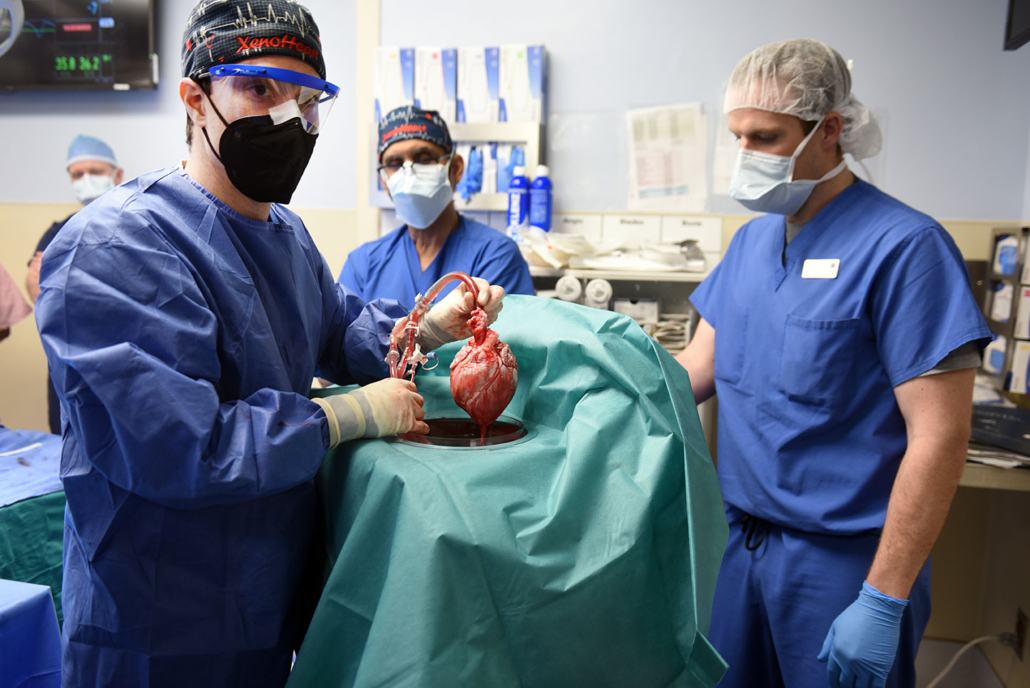
Prior to transplant, the heart from the genetically modified pig is removed from a machine perfusion device to keep it preserved until surgery — University of Maryland School of Medicine
Dr Mohiuddin, Professor of Surgery at UMSOM is considered one of the world’s foremost experts on transplanting animal organs, known as xenotransplantation. He established the Xenotransplantation Program five years ago with Dr. Griffith at UMSOM.
“This is the culmination of years of highly complicated research to hone this technique in animals with survival times that have reached beyond nine months. The FDA used our data and data on the experimental pig to authorize the transplant in an end-stage heart disease patient who had no other treatment options,” Dr Mohiuddin said. “The successful procedure provided valuable information to help the medical community improve this potentially life-saving method in future patients.”
Genetically modified pig
The genetically modified pig was provided by Revivicor, a regenerative medicine company based in Blacksburg, Virginia, US.
Three genes – responsible for rapid antibody-mediated rejection of pig organs by humans – were knocked out in the donor pig. Six human genes responsible for immune acceptance of the pig heart were inserted into the genome. Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue.
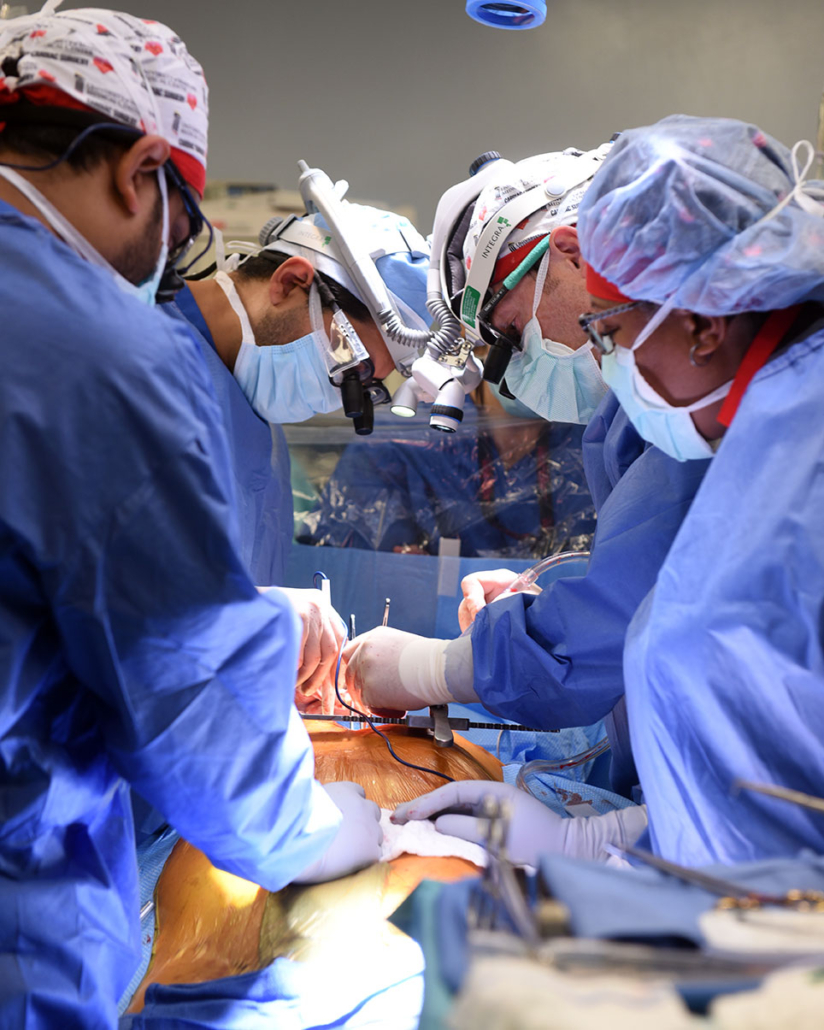
The transplant surgical team was lead by Bartley P. Griffith, MD, and Muhammad M. Mohiuddin, MD. — University of Maryland School of Medicine
Dr. Mohiuddin, Dr. Griffith, and their research team have spent the past five years perfecting the surgical technique for transplantation of pig hearts into non-human primates.
“As a cardiothoracic surgeon who does lung transplants, this is an amazing moment in the history of our field. Decades of research here at Maryland and elsewhere have gone into this achievement. This has the potential to revolutionize the field of transplantation by eventually eliminating the organ shortage crisis,” said Christine Lau, MD, MBA the Dr. Robert W. Buxton Professor and Chair of the Department of Surgery at UMSOM and Surgeon-in-Chief at UMMC. “This is a continuation of steps to making xenotransplantation a life-saving reality for patients in need.”
We can’t give you a human heart
The New York Times quoted Dr. Griffith as saying he first broached the experimental treatment in mid-December. It was a “memorable” and “pretty strange” conversation.
“I said, ‘We can’t give you a human heart; you don’t qualify. But maybe we can use one from an animal, a pig,” Dr. Griffith recalled. “It’s never been done before, but we think we can do it.’”
“I wasn’t sure he was understanding me,” Dr. Griffith added. “Then he said, ‘Well, will I oink?’”
Organs from genetically modified pigs have been the focus of much of the research in xenotransplantation, in part because of physiologic similarities between pigs, human, and nonhuman primates.
UMSOM received $15.7 million sponsored research grant to evaluate Revivicor genetically-modified pig ‘UHearts’ in baboon studies.
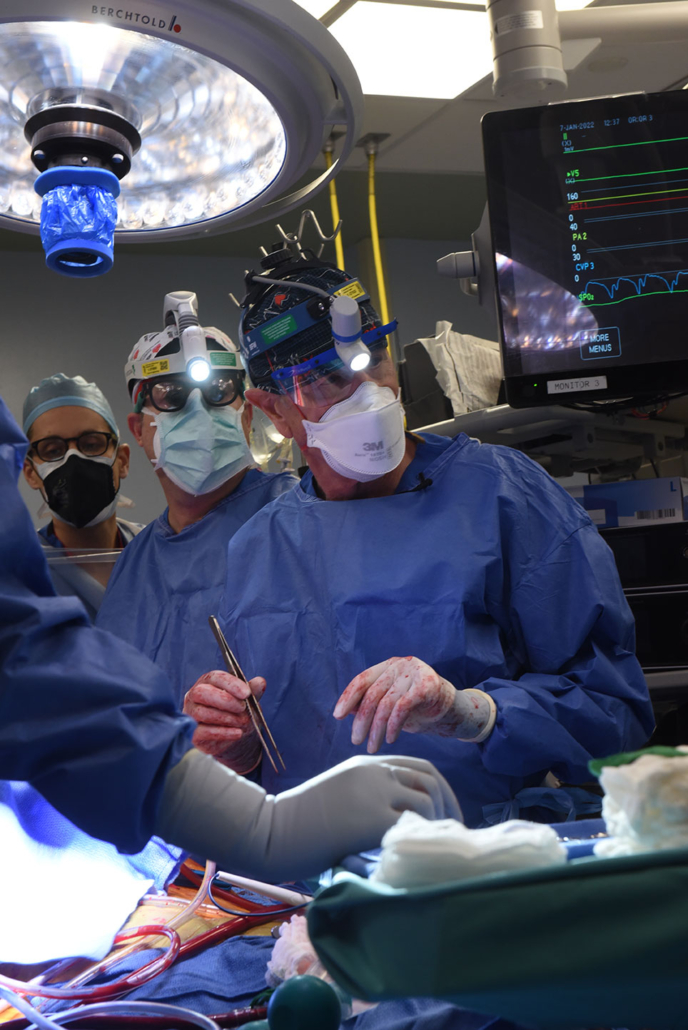
Bartley Griffith, MD, performs the historic transplant — University of Maryland School of Medicine
A new experimental drug made by Kiniksa Pharmaceuticals, along with conventional anti-rejection drugs, which are designed to suppress the immune system and prevent the body from rejecting the foreign organ, were used in the procedure.
Bruce Jarrell, MD, President of the University of Maryland, Baltimore, who himself is a transplant surgeon, recalled: “Dr. Griffith and I began as organ transplant surgeons when it was in its infancy. Back then, it was the dream of every transplant surgeon, myself included, to achieve xenotransplantation and it is now personally gratifying to me to see this long-sought goal clearly in view. It is a spectacular achievement.”