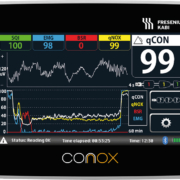
Depth of anesthesia monitor
Fresenius Kabi, a healthcare company that specializes in medicines and technologies for infusion, transfusion and clinical nutrition, has received the CE mark for the Conox anesthesia monitor. Conox is a non-invasive depth of anesthesia monitor designed to help anesthesiologists monitor patient brain activity and to rapidly detect how anesthetics are affecting the patient. The Conox anesthesia monitor comprises cutting edge hardware and digital signal processing technology to measure the two main components of anesthesia: the depth of anesthesia and the patient’s reaction to external stimuli. The qCON index of hypnotic effect and the qNOX index for responsiveness are derived from one single forehead sensor recording the EEG. The Conox monitor features an innovative design with easy to use graphical interface, touch screen display and easy to apply sensors. Conox was developed by Barcelona-based Quantium Medical S.L.U., which was acquired by Fresenius Kabi last year. Quantium Medical is focused on the development of non-invasive physiological monitoring and biosignal data processing. Quantium Medical’s non-invasive monitoring competence is an excellent fit for Fresenius Kabi with its anesthetics I.V. drugs and infusion technology. The new Conox device will provide high-quality anesthesia monitoring and enhance the company’s offering in the operating room and intensive care units.