3D medical printing – the promise of personalization
Three-dimensional (3D) printing for medical applications has grown in recent years at a feverish pace. The technology has long made a significant impact in manufacturing and is also revolutionizing healthcare. For some of its proponents, this would be rather like the Gutenberg printing press did with publishing. Indeed, the respected Gartner Group estimates that 30percent of internal medical implants and devices will be 3D printed by 2020.
3D printing was founded in the 1980s as stereo-lithography’ (STL) and the first commercial 3D printer came to market in 1988. Since the 1990s, manufacturers have used the technique principally for rapid prototyping, or the production of models and moulds.
Implants and prosthetics
Medical 3D printing took off in the early 2000s for producing dental implants and custom prosthetics, with a rapid pace of acceptance in areas such as hearing aids and dental braces. Currently, almost all hearing aids fitted into the ear in industrialiZed countries are made with 3D printers and orthodontic braces, too, are almost entirely 3D printed.
CustomiZed 3D-printed prosthetics and implants were made possible by translation of CT and MRI scans into digital STL print files, and imaging continues to play a central role in medical 3D printing.
Orthopedics and neurosurgery applications
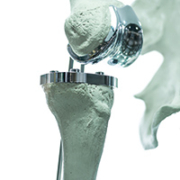
The customization offered by 3D printing also quickly made its case for orthopedic patients being fitted with a standardized hip or spinal prostheses, which required the cumbersome process of shaving of pieces of metal and plastic with scalpels and drills afterwards, in order to achieve best fit.
Neurosurgeons too quickly saw the potential of 3D printing to address the drawbacks of variation in skull shape and the difficulties in using standard cranial implants. In head injury victims, for example, it is important to remove bone to provide space for the brain room to swell and the cranial plate must be perfect in fit.
In situ, in the OR
In the operating room, 3D printing has thoroughly transformed the manufacture of patient models to facilitate planning of surgical procedures. In 2016, a 3D printed model was used by Blythedale Children’s Hospital in Westchester in a 27-hour operation to separate twins conjoined at the head. According to many reports, recovery of the infants was accelerated due to the 3D model.
At the University of Michigan, CT images of a patient’s airway were used with a 3D printer to fabricate a precisely modelled, bioresorbable tracheal splint that was surgically implanted in a baby. The baby recovered, and full resorption of the splint is expected to occur within three years.
3D printing has also been used for making surgical tools such as forceps, hemostats, scalpel handles and clamps. They are formed sterile, and some estimates report that they cost only a tenth of a stainless steel equivalent.
In situ printing, by which implants, tissue (and eventually organs) are 3D printed in the human body during operations is anticipated in the future trend. Such a trend is being reinforced by rapid developments in miniaturized robotic bioprinters and robot-assisted surgery.
Personalized pills
3D printing technologies are also used for personalized medicine, with precision in dose (matched to patient profile and response). Some firms are experimenting with complex drug-release profiles, such as poly-pills with multiple active ingredients in a multilayered form.
This is seen as promising new standards of care for patients with several chronic diseases. Extended to one poly-pill per day for everyday medications, such a step would reduce a bane of medical practitioners – namely patient non-compliance.
In 2016, Spritam levetiracetam, a new drug to control seizures brought on by epilepsy, was approved by the US Food and Drug Administration (FDA). The pill, the world’s first to be 3D printed, is based on a trademarked ZipDose technology developed by Ohio-based Aprecia, and provides more porosity than alternative dosage forms.
Industry experts foresee drugs manufacturing being done eventually at the point-of-care, with physicians emailing medication formulations to pharmacies for on-demand drug printing.
Post-industrial production
The logic of 3D printing is in some ways truly revolutionary. What it brings is an end to the idea that there is commercial sense in only large runs of standardized products, a cornerstone of 19th/20th century manufacturing tradition as well as the Industrial Revolution. The first 3D manufactured product, in other words, costs approximately the same as the next one.
3D printing also reduces cost in certain cases. For example, a 5-mg pharmaceutical tablet can be custom-fabricated on demand as a smaller and less expensive 2.5-mg tablet rather than being broken up and left unused.
Speed
Speed too is a major asset of medical 3D manufacturing, and a spin-off from the fact that large production runs are not required. Customized products like prosthetics and implants, in particular, can be made within hours.
As with pharmacy pills, some expect on-site 3D printing at, or adjacent to, a hospital, to eventually emerge, for making patient-specific products.
Basic technology
The basic technique of 3D printing, which is also known as additive manufacturing, involves the successive deposit of layers of materials, typically plastic and ceramics or metal and powders, to make the final product.
One of the most exciting innovations, however, consists of using live cells as the printing material.
Types of 3D printer
The type of 3D printer chosen for an application often depends on the material used and the method for bonding the layers in the final product. Key technologies for medical applications include selective laser sintering (SLS) and thermal inkjet (TIJ) printing. Another widely-used 3D printing technology is fused deposition modelling (FDM).
Though relatively basic and inexpensive, FDM was one of the earliest examples of successful medical 3D printing in the late 1990s/early 2000s when it was used to construct cranial implants. FDM remains widely used for rapid modelling and prototyping in orthopedics and dentistry.
FDM printers use a print-head similar to an inkjet printer. Rather than ink, however, beads of thermoplastic (similar to those used in injection moulding) are released to form a thin layer. The process is repeated continuously. Since the plastic is heated, it fuses to the layers below, and then hardens as it cools to create the final product.
More complex medical uses of 3D printing are based on SLS and TIJ.
SLS uses metal, plastic or ceramics as material. A laser draws out the shape of the object and this is then fused to a powdered metal substrate. The process is repeated until the product is formed. The degree of detail in SLS is directly linked to the precision of the laser and the powder’s fineness.
On its part, TIJ uses thermal (as well as electromagnetic or piezoelectric) technology to deposit tiny droplets of ink or even cells (bio-ink) on a substrate. Unlike office inkjet printers, 3D TIJ heats a print-head to create collapsing air bubbles, which in turn create pressure pulses to eject the droplets from nozzles. The size of the droplets can be adjusted by temperature, pulse frequency or material viscosity and volumes can be as little as 10-20 picolitres. Multiple-head TIJ is especially promising for producing tissue and simple organs in the process of bioprinting’ (discussed below). Other applications under study include drug delivery and gene transfection.
Bioprinting – the final frontier
While implants and prosthetics have convincingly demonstrated the real-world relevance of 3D printing, the maximum excitement is currently focused on its use in tissue and organ fabrication.
Ageing, accidents, disease and birth problems often cause tissue and organ failure. Treatment is largely based on donor transplants. However, there is a chronic shortage of supply, not least of suitable donors (e.g. with matching tissue). In addition, surgery and follow-up is complex and expensive.
One recent approach to finding a solution consists of tissue engineering and regenerative medicine, based on mixing growth factors into isolated stem cells, multiplying them in a lab and then seeding the cells on scaffolds which transform direct cell proliferation and differentiation into functioning tissues.
Beyond regenerative medicine
Bioprinting takes traditional regenerative technologies further than scaffold support alone by using 3D printing technology to produce layers of cells, biomaterials, and cell-laden biomaterials. This is then precisely placed by the printer in tissue-like structures. As mentioned previously, inkjet-based bioprinting is the most commonly used technique for bioprinting.
Tissues and organs
German researchers have been developing skin cell bioprinting since 2010. In January 2017, a team from Spain’s Universidad Carlos III de Madrid (UC3M) reported in the journal Biofabrication’ they had developed 3D-printed human skin adequate for transplant into patients, and for testing drugs and cosmetics. Their product is currently undergoing European approval. Meanwhile, in the US, Organovo too has developed 3D-printed skin. Demonstrating the potential of such markets, French cosmetics giant L’Oreal has begun collaborating with Organovo.
Researchers have so far also successfully printed a knee meniscus, heart valves, bone and an artificial liver. In 2016, scientists at Cambridge University’s Centre for Brain Repair reported the 3D printing of a retina using a piezoelectric TIJ printer.
One application area is to use 3D printing to create tissues and organs for medical research, and rapidly screen candidate drugs, cutting research costs and time. Organovo is developing strips of printed kidney and liver tissue for exactly such a purpose, while Russia’s 3D Bioprinting Solution has 3D printed a functional thyroid in a mouse and claims to be ready to do the same in humans.
20 years to a 3D-printed heart?
Nevertheless, most bio-printed organs have so far been relatively small and simple, with no vascularity or nerve system and nourishment provided wholly by diffusion from the host vasculature. Such diffusion seems to suffice for thicknesses of 150-200 micrometers. Beyond it, there is none. In future, the bioprinting of 3D organs such as an entire kidney or heart will require precise multicellular structures with full vascular network integration.
Such a process may not be that far away. Collaborators from a network of academic institutions, including the Harvard University, Stanford University, the Massachusetts Institute of Technology and the University of Sydney recently announced they had bioprinted a perfusable network of capillaries, marking a significant stride toward overcoming the limits to diffusion.
According to some projections, we may be less than 20 years from a fully functioning printable heart.
Challenges ahead
As with many other frontiers of medicine, an immediate challenge for medical 3D printing consists of regulatory acceptance. Though a hundred-odd 3D-printed products had been approved in the US and Europe by the end of 2016, these consist almost entirely of prosthetics, surgical tools and artificial bone replacement.
Fulfilling regulatory requirements for more complex products is likely to be much more demanding. Included here are the need for large randomized controlled trials, which require funding and time – for instance to determine the biocompatibility of several of the new materials being used.