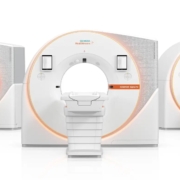
Siemens expands photon-counting CT portfolio with new class of scanners
Three years after launching the world’s first photon-counting computed tomography scanner, Siemens Healthineers has expanded its portfolio with two new systems: the dual-source Naeotom Alpha.Pro and the single-source Naeotom Alpha.Prime. These innovations promise enhanced diagnostic capabilities through superior image quality at lower radiation doses, with potential to change clinical pathways. Callan Emery attended the launch […]