ScreenPoint Medical Hits New Milestones
World’s Leading Breast AI Software, Transpara Surpasses 5 Million Mammograms and 1 Million Tomosynthesis Studies Analyzed to Support Radiologists Reading Mammograms
ScreenPoint Medical announced that its Transpara® breast AI has surpassed 5 million mammograms, including over 1 Million Tomosynthesis (3D) exams analyzed in support of radiologists reading mammography exams. Transpara provides radiologists with a ‘second pair’ of eyes helping detect cancers earlier and reduce recall rates.
Used by hundreds of sites globally, Transpara has set the standard for Breast AI, supporting all major mammography gantry manufacturers, and integrating into radiology reading workflow both in the single-reading and double-reading environments. ScreenPoint will showcase its industry leading Transpara results at the upcoming 109th Annual Radiological Society of North America (RSNA) meeting, November 26-30, 2023 (South Hall #3947).
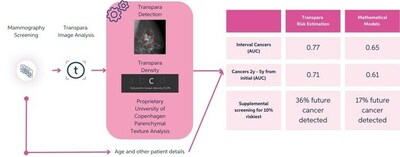
The clinical and workflow benefits of Transpara on improving the mammography screening process have been the subject of several major research studies published in top-tier peer-reviewed publications throughout the year, including a recent Radiology publication (Lauritzen, et al) in August demonstrating that Transpara combined with two other measures delivered a superior short term risk measure relative to many current models (i.e., Gail, TC8). (see attached figure)
“As the most widely utilized Breast AI in clinical practice with 5 million mammograms and 1 million tomosynthesis exams analyzed, we are very proud that study after study continues to demonstrate the value that Transpara brings to breast health care provider and patients. Regardless of country, patient age, breast density, or patient ethnic background, Transpara delivers consistent results. Our in-development offerings will continue to expand the reach and impact of Transpara Portfolio in helping radiologists detect cancer earlier and safely reduce workload,” said Mark Koeniguer, ScreenPoint Medical CEO.
Transpara is FDA cleared and has European regulatory approval (CE Mark) for use with 2D and 3D mammography from multiple manufacturers. Used by hundreds of leading centers in more than 30 countries, Transpara is designed to work concurrently with radiologists. Research shows that up to 45% of interval cancers can be found earlier using Transpara, while helping to reduce workload and optimize workflow.